- Adhera enters licensing agreement for Parkinson’s drug candidate…
- Treatment uses compound previously approved in Europe…
- Company plans to fast-track clinical trials using previous safety data…
Adhera Therapeutics Inc (OTCMKTS: ATRX) is soaring on news of a licensing agreement for a new Parkinson’s drug candidate.
ATRX stock popped 360% higher early in the session and was holding more than 160% higher at writing.
The rally comes after the biopharmaceutical company announced it has executed a definitive license agreement with Melior Pharmaceuticals II, LLC for the initial purpose of developing a new therapeutic for Parkinson’s disease.
Table of Contents
Details of The Deal
The agreement is for the development of Melior 2’s drug candidate MLR-1019 which is an armesocarb treatment.
Adhera said armesocarb is the active enantiomer in mesocarb, which was a drug marketed in Europe for the treatment of psychiatric and central nervous system indications.
The company said “the Russian manufacturer of mesocarb discontinued operations for reasons unrelated to the compound” in 2008.
“The compound has a well-documented safety profile undergirded by use in more than one million patients across a wide therapeutic dosing range during its time on the market.”
Adhera and Melior 2 expect to leverage existing safety data from that drug to advance MLR-1019 directly into a Phase 2a clinical trial to approve the drug for Parkinson’s treatment.
Adhera said the treatment “will be the only drug candidate in development that addresses both movement (e.g., facial masking, dyskinesia, etc.) and non-movement (e.g., sleep disorders, cognitive deficits, etc.) symptoms of Parkinson’s.”

Image credit: Kotcha K/Shutterstock.com
Melior secured several patents for the intellectual property on MLR-1019 after discovering the benefits of the drug for Parkinson’s treatment during research.
The two companies said they believe the drug “represents a significant opportunity to address a large gap in therapeutics for PD.”
Melior 2 believes the drug can increase the effectiveness of the drug levodopa, which is a drug commonly prescribed for controlling movement symptoms of Parkinson’s.

Image credit: Sonis Photography/Shutterstock.com
They also believe MLR-1019 can mitigate the dyskinesia symptoms associated with that therapy “more effectively than amantadine and without the unpleasant adverse events.”
Adhera CEO, Andrew Kucharchuk, said, “We are thrilled to have the Melior group of companies as partners and collaborators on this project and look forward to evaluating MLR-1019 in a clinical setting with the goal of delivering a safe and effective option to patients and neurologists to better manage this debilitating disease.”
Andrew Reaume, CEO of Melior, said, “We are delighted with the Agreement and to be working with the Adhera team, as we have a shared vision towards armesocarb as an innovative new therapy to address multiple aspects of Parkinson’s disease. Our full set of global resources are now at the disposal of Adhera and we look forward to deepening our relationship based upon the success of our work in PD.”
Under the agreement, “Adhera will be granted exclusive worldwide rights to the MLR-1019 IP and Melior know how to develop MLR-1019 in exchange for payments that are only triggered by successful completion of specific clinical trial and FDA approval milestones.”
If the drug is approved, “Melior will also be owed a royalty on all future gross product sales.
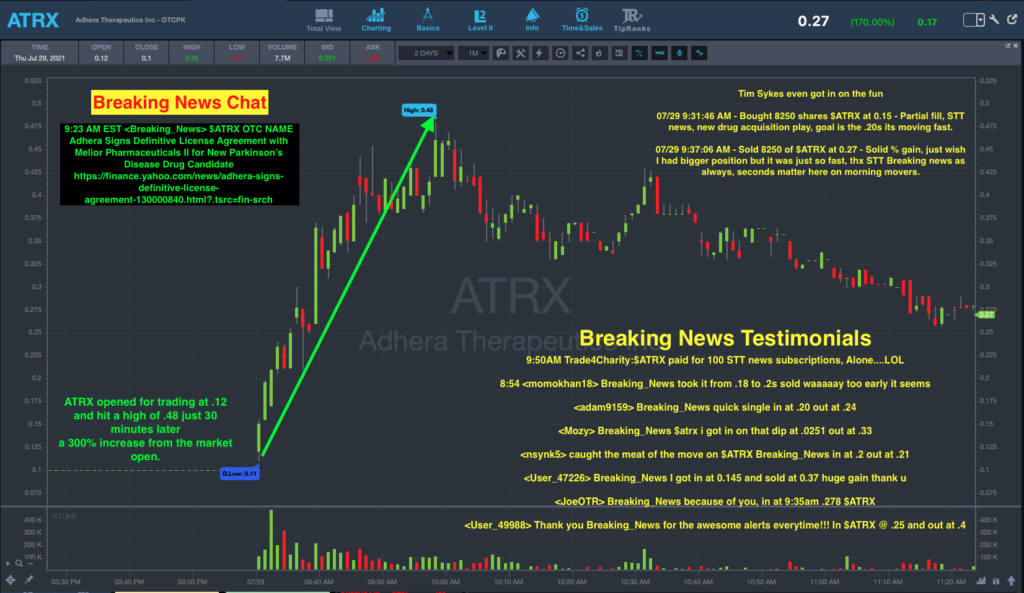
STT Breaking News Chat Alerts On ATRX Stock
If you’re a subscriber to the StocksToTrade Breaking News Chat, you may have seen this news about ATRX stock before the market open and been able to get in on the rally.
An alert about the company’s press release was sent in the chat ahead of the market open at 09:23 a.m. Eastern.
The stock then opened trading at $0.15 per share before topping $0.48 per share by 10:00 AM.
Self-made trading millionaire, philanthropist, and educator Tim Sykes reminded his Twitter followers that alerts like this are why the STT platform is so key.
https://t.co/Jypm5k0x1a This is why STT Breaking News is a MUST use every single dam,n day, $ATRX
— Timothy Sykes (@timothysykes) July 29, 2021
Closer Look at ATRX Stock
- Adhera Therapeutics was founded in 1983 and is headquartered in North Carolina.
- The company describes itself as “a clinical-stage biopharmaceutical company focused on identifying advanced drug candidates that may qualify for accelerated developmental pathways.”
- Adhera’s “legacy assets include CEQ508, an oral delivery of small interfering RNA (siRNA) against beta-catenin, to suppress polyps in the precancerous syndrome and orphan indication Familial Adenomatous Polyposis (FAP).”
- The stock closed at $0.10 on Wednesday and hit a daily high of $0.48 today.
About Melior
- Melior Discovery was founded in 2005 and is headquartered in Exton, PA.
- Melior Discovery and Melior Pharmaceuticals are privately held.
- Melior Discovery is the parent company of Melior Pharmaceuticals I, Inc. and Melior Pharmaceuticals II, LLC.
- Those two companies are “leaders in pharmaceutical drug repositioning using the unique theraTRACE platform comprised of multiplexed in vivo disease models.”
Featured cover image credit: Joyseulay/Shutterstock.com